Page 45 - 1.HTAIn_Process_Manual
P. 45
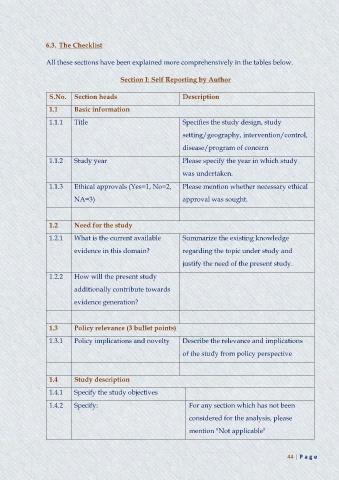
6.3. The Checklist
All these sections have been explained more comprehensively in the tables below.
Section I: Self Reporting by Author
S.No. Section heads Description
1.1 Basic information
1.1.1 Title Specifies the study design, study
setting/geography, intervention/control,
disease/program of concern
1.1.2 Study year Please specify the year in which study
was undertaken.
1.1.3 Ethical approvals (Yes=1, No=2, Please mention whether necessary ethical
NA=3) approval was sought.
1.2 Need for the study
1.2.1 What is the current available Summarize the existing knowledge
evidence in this domain? regarding the topic under study and
justify the need of the present study.
1.2.2 How will the present study
additionally contribute towards
evidence generation?
1.3 Policy relevance (3 bullet points)
1.3.1 Policy implications and novelty Describe the relevance and implications
of the study from policy perspective
1.4 Study description
1.4.1 Specify the study objectives
1.4.2 Specify: For any section which has not been
considered for the analysis, please
mention "Not applicable"
44 | P a g e