Page 47 - Htain Manual
P. 47
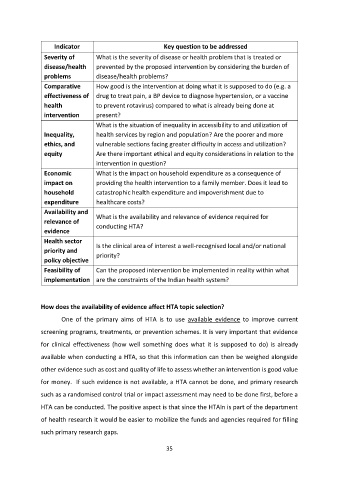
Indicator Key question to be addressed
Severity of What is the severity of disease or health problem that is treated or
disease/health prevented by the proposed intervention by considering the burden of
problems disease/health problems?
Comparative How good is the intervention at doing what it is supposed to do (e.g. a
effectiveness of drug to treat pain, a BP device to diagnose hypertension, or a vaccine
health to prevent rotavirus) compared to what is already being done at
intervention present?
What is the situation of inequality in accessibility to and utilization of
Inequality, health services by region and population? Are the poorer and more
ethics, and vulnerable sections facing greater difficulty in access and utilization?
equity Are there important ethical and equity considerations in relation to the
intervention in question?
Economic What is the impact on household expenditure as a consequence of
impact on providing the health intervention to a family member. Does it lead to
household catastrophic health expenditure and impoverishment due to
expenditure healthcare costs?
Availability and
What is the availability and relevance of evidence required for
relevance of
conducting HTA?
evidence
Health sector
Is the clinical area of interest a well-recognised local and/or national
priority and
priority?
policy objective
Feasibility of Can the proposed intervention be implemented in reality within what
implementation are the constraints of the Indian health system?
How does the availability of evidence affect HTA topic selection?
One of the primary aims of HTA is to use available evidence to improve current
screening programs, treatments, or prevention schemes. It is very important that evidence
for clinical effectiveness (how well something does what it is supposed to do) is already
available when conducting a HTA, so that this information can then be weighed alongside
other evidence such as cost and quality of life to assess whether an intervention is good value
for money. If such evidence is not available, a HTA cannot be done, and primary research
such as a randomised control trial or impact assessment may need to be done first, before a
HTA can be conducted. The positive aspect is that since the HTAIn is part of the department
of health research it would be easier to mobilize the funds and agencies required for filling
such primary research gaps.
35